Laparoscopic Splenectomy
What is the spleen?
The spleen is an organ situated in the left upper abdomen and plays a major role in body defence against infections.
When is a splenectomy advisable?
Splenectomy i.e. removal of the spleen may be advisable in patients who suffer from some bleeding disorders e.g. in Idiopathic thrombocytopenic purpura, thrombotic thrombocytopenic purpura or from haemolytic anaemia. The spleen can be removed either at open surgery or a laparoscopic surgery.
Why should I undergo a laparoscopic splenectomy?
There is less pain with an earlier return of bowel function. There is usually less bleeding than in open splenectomies. Hospital stay is also shorter, which varies from 2 to 5 days after laparoscopy as opposed to 5 to 9 days after traditional open surgery.
What happens at the first appointment?
Your surgeon will initially take a history and examine you. He will also ensure that you are fit to undergo surgery and an anaesthetic. If you are less than 40 year old and generally very fit then he may suggest an operation without further appointments or tests.
What happens at the subsequent appointments?
Your surgeon will check that all necessary information and reports are available and have been evaluated. He will discuss the results and reports of various investigations. He will discuss with you the various options available to treat your hernia. If you are willing to undergo surgery then he will agree with you a date for the procedure.
Admission for surgery
You must not eat any food from 6 hours before the operation. You are allowed to drink clear fluids, including tea and coffee without milk up to 3 hours before the operation. A nurse will admit you on the day of surgery. The surgeon and the anaesthetist will see you prior to your operation. They will confirm that you are willing to undergo surgery, all preparations are complete and it is safe to proceed with your operation. They will confirm that an appropriate facility is available for you to recover after surgery.
What happens during surgery?
The operation is performed under general anaesthetic and a small ‘telescope’ (laparoscope) is passed into your abdomen above the navel. Your abdomen is filled with carbon dioxide gas, to help see everything properly. Further 4 small cuts will be made in your abdomen to insert instruments to help with the operation. The surgeon will disconnect the hernia sac from the abdominal wall and then put a mesh in place with metal staples from inside.
What happens in hospital after surgery?
You will be sent to the Ward after the procedure. You will be looked after by a nurse and offered appropriate pain-killers. You will be offered light food and drink. You may be provided with a system that will allow you to self-administer effective painkillers safely (Patient Controlled Analgesia) – do not worry as you can not overdose on this system. This involves setting up a pump to administer small amounts of morphine in to your vein when you press a button in your hand. Alternatively, you may be prescribed oral painkillers with injections for back up. You may feel drowsy and develop constipation as a result of your painkillers - hence it is advisable that you drink plenty of fluids. You should be able to go home 1-3 days after your operation, depending on your home circumstances and your need for pain-killers. You will have a few small wound dressings which you can remove yourself after one week or a nurse / the surgeon can remove these at a subsequent visit.
Support available during early recuperation period after surgery
It is advisable to avoid strenuous physical activity for the first 2 weeks to allow proper healing of the wounds. It is usual for you to experience some initial pain in the wounds and in your abdominal wall - you may also experience minor discomfort in the shoulder. Most patients recover fully within 1-2 months. You will be given a number to ring for advice in case of difficulty.
Further follow up after surgery
You will be given an appointment to see the surgeon again - he will ensure that you are recovering as expected.
Will further surgery be needed?
This is usually not necessary, though rarely surgery may be required if you develop complications or the hernia recurs.
 1. Length of procedure. 1. Length of procedure.
1.1. Statistics
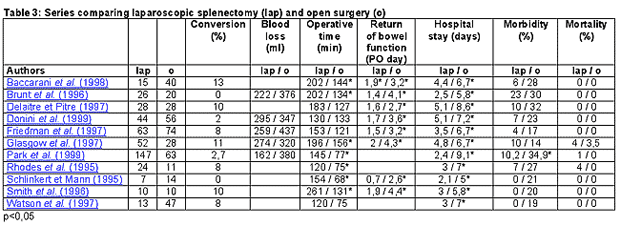
Operative time varies from 65 to 340 minutes, with an average of about 160 minutes ( Emmermann et al. , 1995; Katkhouda et al. , 1998; Delaitre et al. , 2000 ). In these series, the surgeon’s experience influences the results with a significantly shorter operative time after the tenth procedure (p<0.01). Obesity with a body mass index (BMI)>30 significantly increases operative time (p = 0.01). A spleen larger than 15 cm on the preoperative ultrasound evaluation also increases the length of the procedure, which reaches 207 minutes in the series of Katkhouda et al. (1998) . In articles comparing open and laparoscopic surgery, the duration of laparoscopic splenectomy is significantly longer: p<0.05 (table 1.1).
Thrombocytopenia is no longer considered a contraindication for laparoscopic surgery: failure in the preoperative treatment with a platelet count of less than 10 000 was found in 6% of cases in the series of Kathkouda et al. (1998) without any particular consequences. The platelet count was less than 30 000 in 10.5% of patients in the French series ( Delaitre et al. , 2000 ).
Intraoperative platelet transfusion is necessary in these cases ( Katkhouda et al. , 1998 ). Red blood cell transfusion is rarely indicated: 2% to 6% of patients ( Emmermann et al. , 1995; Flowers et al. , 1996; Katkhouda et al. , 1998 ), except when a conversion was required ( Flowers et al. , 1996; Katkhouda et al. , 1998; Kusminsky et al. , 1995 ). There is usually less bleeding than in open splenectomies ( Yee et al. , 1995 ). Auto-transfusion, which we initially performed, no longer seems necessary. Thrombocytopenia even when severe is therefore no longer a contraindication for laparoscopic splenectomy.
In the series comparing laparoscopy and open surgery, there is an earlier return of bowel function, with a significant 50% gain in favor of laparoscopic splenectomies (table 1.1). Pain and the analgesic intake are significantly reduced ( Delaitre and Pitre, 1997; Hashizume et al. , 1996 ). There is also a significant difference regarding the length of hospital stay, which varies from 2.4 to 5 days after laparoscopy as opposed to 5 to 9 days after laparotomy.
Table 1.1 Series comparing laparoscopic splenectomy (lap) and open surgery (o)
1.2. Accessory spleens
Their incidence rate ranges from 9.3% to 27.5% ( Flowers et al. , 1996; Katkhouda et al. , 1998; Park et al. , 1999; Delaitre et al. , 2000; Watson et al. , 1997; Friedman et al. , 1996 ) with an average rate of 15%. This corresponds to the incidence rate for the general population which is between 15% and 20%. In the comparative series (table 1.2), no significant differences are found between laparoscopic splenectomies and open splenectomies. Gigot et al. (1998) was the only series reporting a higher rate of accessory spleens (41%). In this series, scintigraphies performed 24 months after surgery revealed residual splenic tissue in 50% of cases, which were probably due to splenosis caused by an intraoperative rupture of the splenic capsule. This phenomenon is frequent after traumatic lesions of the spleen (44% to 76% of cases ( Gunes et al. , 1994; Nielsen et al. , 1981; Normand et al. , 1993 ). It is between 15% and 20% after open splenectomy for hematological disease ( Spencer et al. , 1981 ). The quality of the dissection, the absence of hemorrhage and the use of a bag for extraction of the spleen are important factors for the prevention of splenosis.
Table 1.2: Accessory spleens: laparoscopy versus open surgery
 2. Conversions. 2. Conversions.
The rate of conversion to laparotomy was between 16% and 20% in the first series reported ( Emmermann et al. , 1995; Flowers et al. , 1996; Phillips et al. , 1994 ). In recent series, it is less than 10% ( Rhodes et al. , 1995; Katkhouda et al. , 1998; Trias et al. , 2000; Park et al. , 1999; Donini et al. , 1999; Friedman et al. , 1996; Smith et al. , 1996 ), or even nil ( Brunt et al. , 1996; Schlinkert and Mann, 1995; Tsiotos and Schlinkert, 1997 ). We have had to convert to open splenectomy once during the last 40 splenectomies performed in our services for idiopathic thrombopenic purpura or hemolytic anemia.
The majority of conversions are due to hemorrhaging involving the splenic pedicle, the short gastric vessels, the spleen or the large neighboring vessels [2/3 of the conversions in the French series ( Delaitre et al. , 2000 )]. The other causes are related to difficulties in gaining access to the pedicle because of obesity or extensive adhesions.
In the French study ( Delaitre et al. , 2000 ), the conversion rate is significantly correlated to the following factors:
- Obesity (BMI>30: p<0.001)3
- Hemostasis technique, which is significantly better using stapling than ligature or clips (p<0.001)
- Splenomegaly (weight of the spleen>350g: p<0.05)
- Surgeons’ experience, based on 10 splenectomies (p<0.001)
Splenomegaly is a major cause of operative difficulty. In the series of Targarona et al. (1998) , 3 conversions were required out of 19 cases (15.8%) of massive splenomegaly (weight>2500g) due to the lack of space for manipulating the instruments. Schlachta et al. (1999) also reported 3 conversions (21%) for splenectomies performed for malignant blood disease involving enlarged spleens (23, 24 and 27 cm). More moderate splenomegalies (400 g to 2000 g) are no longer considered a contraindication for laparoscopic surgery ( Targarona et al. , 1998 ).
A comparison between conversions and purely laparoscopic procedures ( Delaitre and Pitre, 1997 ) reveals that conversion, significantly delays the return of bowel function (62.9 hours versus 38.5 hours: p<0.001); prolongs the duration of analgesic intake (3.9 days versus 2.3 days) and prolongs the mean hospital stay (10.1 days versus 5.5 days (p<0.001) due, notably, to greater morbidity (27.3% versus 10.4%: p<0.002).
3. Complications.
The frequency of complications varies from 0 to 23% (Table 1.1).
3.1. Morbidity
In the French series ( Delaitre et al. , 2000 ), there was an overall morbidity of 13.8% (n = 38). It was significantly higher in case of conversion (27.3% vs. 10.4%: p<0.002), obesity (20% vs. 7.7%: p<0.001) and malignant disease (33% vs. 12.6%: p<0.001). If the results of 12 series are combined, out of a total of 795 laparoscopic splenectomies (Table 3.1: Emmermann et al. , 1995; Flowers et al. , 1996; Rhodes et al. , 1995; Katkhouda et al. , 1998; Trias et al. , 2000; Park et al. , 1999; Delaitre et al. , 2000; Donini et al. , 1999; Glasgow et al. , 1997; Smith et al. , 1996; Watson et al. , 1997; Hashizume et al. , 1996 ), a 9% rate of complications is observed (72 cases). The most frequent types of complications are either parietal (hematomas, abscesses, exenteration), intraperitoneal hemorrhaging, pneumopathies and pleurisies, subphrenic abscesses or hematomas. Three pulmonary embolisms occurred as a result of phlebitis (5 cases). Postoperative acute pancreatitis was also reported ( Gigot et al. , 1996; Katkhouda et al. , 1998; Delaitre et al. , 2000; Donini et al. , 1999; Tsiotos and Schlinkert, 1997 ). Indeed, the tail of the pancreas is situated less than 1 cm from the splenic hilum in 30% of cases (75% of patients). Dissection of the splenic vessels and of the tail of the pancreas must therefore be performed attentively and with particular caution. This maneuver is facilitated by the “hanging spleen” technique during which the spleen remains attached to the diaphragm and to the pancreas, and the splenic vessels are tented ( Katkhouda et al. , 1998; Delaitre, 1995 ). Thirteen re-operations were required (18%).
Table 1.3: Complications of laparoscopic splenectomies (12 series: Emmermann et al. , 1995; Flowers et al. , 1996; Rhodes et al. , 1995; Katkhouda et al. , 1998; Trias et al. , 2000; Park et al. , 1999; Delaitre et al. , 2000; Donini et al. , 1999; Glasgow et al. , 1997; Smith et al. , 1996; Watson et al. , 1997; Hashizume et al. , 1996 )

| Parietal complications |
14 |
| Intraperitoneal hemorrhaging/bleeding |
13 |
| Pneumopathies – Pleurisies |
11 |
| Subphrenic collection |
9 |
| Thrombophlebitis/Pulmonary embolism |
5 |
| Ileus – Impaction |
4 |
| Pancreatitis |
4 |
| Anemia |
2 |
| Cardiac complication |
1 |
| Urinary complication |
1 |
| Others |
6 |
| |
72/795 (9%) |
In the series comparing laparoscopy to laparotomy, complications are less frequent after laparoscopy (table 1.1) and they are usually less serious ( Katkhouda et al. , 1998 ).
In the series comparing splenectomies for hematologic malignancies and benign disease, the complications observed were similar for the two groups for Schlachta et al. (1999) . However, most authors agree that complications are more frequent for a hematologic malignancy ( Delaitre et al. , 2000; Klingler et al. , 1999; Horowitz et al. , 1996; Schwartz, 1996 ). HIV-related disease does not seem to worsen the immediate prognosis: 1 complication out of 8 cases reported by Trias et al. (2000).
3.2. Mortality
There were no intraoperative deaths reported by the literature. Perioperative mortality is nil or low for laparoscopic splenectomies (0 to 4%) whereas it varies from 1% to 9% for open splenectomies ( Horowitz et al. , 1996; Schwartz, 1996; Musser et al. , 1984 ). Perioperative mortality in laparoscopic splenectomies for benign hematologic disease is usually nil or very low; three cases are reported by the literature: sepsis secondary to a central catheter in a patient with ITP and pulmonary hypertension leading to death on POD8 ( Flowers et al. , 1996 ); an intraoperative and postoperative hemorrhage in a patient undergoing surgery for auto-immune hemolytic anemia and suffering from cardiac failure with death on POD33 ( Schlachta et al. , 1999 ); 1 death on POD8 due to thrombosis of the inferior vena cava and superior mesenteric vein after splenectomy for hypersplenism ( Park et al. , 1999 ). In thrombotic thrombocytopenic purpura (TTP), Glasgow et al. (1997) reported 2 deaths due to complications of preexisting nervous system involvement.
In malignant disease, deaths are not frequent (0 to 7%): 1 death was reported on POD7 (4.2%) due to a myocardial infarction in a patient with a lymphoma ( Rhodes et al. , 1995 ), 1 death on POD28 (4.7%) due to a pneumopathy in a patient with chronic lymphocytic leukemia ( Flowers et al. , 1996 ), 1 death (7%) due to pancreatitis and sepsis in a HIV-positive patient with a Hodgkin’s lymphoma ( Schlachta et al. , 1999 ).
3.3. Malignant hematological disorders
There is a higher conversion rate than for benign disorders (21% for Schlachta et al. , 1999 ), related to the size of the spleen and frequent adenopathies in the splenic pedicle region. The series of Donini et al. (1999) does not report a significant difference in morbidity for laparoscopic splenectomies performed for ITP as opposed to those performed for malignant hemopathies, whereas the French series ( Delaitre et al. , 2000 ) reports a higher rate of morbidity for malignant hemopathies: 33% vs. 12.6%. In one series comparing laparoscopy and open surgery ( Schlachta et al. , 1999 ), the morbidity rates were not statistically different: 9% and 11% with one death following laparoscopy. For Donini et al. (1999) , however, the morbidity rate is lower for laparoscopy than for open surgery: 7.7% vs. 23.8%. Trias, in his series of 28 cases ( Trias et al. , 2000 ), reported a morbidity of 28% with 2 hemorrhagic complications. These figures are much lower than those of Horowitz et al. (1996) who reported a morbidity rate of 52% and a mortality rate of 9% for 135 open splenectomies with a significantly higher rate of complications when the spleen weighed more than 2 kg.
 4. Long-term results for benign disorders. 4. Long-term results for benign disorders.
4.1. Idiopathic thrombocytopenic purpura
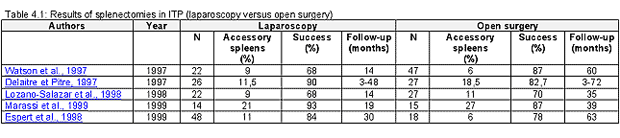
In the few studies which report long-term results of splenectomies, the average follow-up period ranges from 14 to 48 months ( Katkhouda et al. , 1998; Trias et al. , 2000; Delaitre and Pitre, 1997; Tsiotos and Schlinkert, 1997; Zamir et al. , 1996 ) (Table 4.1). There is a favorable outcome in over 80% of patients ( Katkhouda et al. , 1998; Trias et al. , 2000; Delaitre et al. , 2000; Friedman et al. , 1997; Watson et al. , 1997; Tsiotos and Schlinkert, 1997; Zamir et al. , 1996; Szold et al. , 2000 ). These figures are comparable to those obtained for open surgery ( Delaitre and Pitre, 1997; Watson et al. , 1997 ). At present, the success rate of splenectomies is considered to be over 90% if the platelet destruction after splenectomy is fully splenic, and 62% when the destruction is mixed (hepatic and splenic) ( Najean et al. , 1997 ). A sharp rise in the platelet count during the postoperative period is a factor of good prognosis: Katkhouda et al. (1998) observed no recurrences in patients whose platelet count was over 160 000/mm3 during the first 3 days following surgery. These findings were underscored by Julia et al. (1990) in a multivariate study of treatment response factors. These recurrences can be the result of an extra-splenic platelet destruction: medullary, hepatic or in an accessory spleen ( Gigot et al. , 1998 ). For some authors, accessory spleens are responsible for 70% of recurrences ( Rudowski, 1985 ). The presence of residual splenic tissue (splenosis or accessory spleen revealed by postoperative scintigraphy) varies from 12% ( Facon et al. , 1992 ) to 48% ( Akwari et al. , 1987 ). Gigot et al. (1998) reported a 50% presence of residual splenic tissue 24 months after surgery, with a recurrence rate of 16%. The growth of accessory spleens is slow, however. Several years may be required before an effect on the thrombocytopenia can be noticed. The removal of accessory spleens, which leads to an improvement in the clinical course in 27% to 75% of cases ( Akwari et al. , 1987; Verheyden et al. , 1978 ) is often possible using a laparoscopic approach ( Watson et al. , 1997; Morris et al. , 1999; Szold et al. , 2000 ). Recurrences are also observed in cases of associated pathologies such as lupus erythematosus ( Trias et al. , 2000 ).
Results of splenectomies in ITP (laparoscopy versus open surgery)
4.2. Thrombotic thrombocytopenic purpura
The results are less favorable: 4 successful outcomes out of 6 patients in the series of Katkhouda et al. (1998) , but 6 failures out of 6 cases in the series of Bell et al. (1991) . The limited number of patients operated on do not enable to determine predictive factors for success.
4.3. Hemolytic anemia
The results are excellent in spherocytoses: 90% to 100% success rates after follow-up of 27 to 37 months ( Katkhouda et al. , 1998; Trias et al. , 2000 ). In auto-immune anemias, laparoscopic splenectomy obtains a durably successful outcome in respectively 70%, 88% et 92% of cases ( Katkhouda et al. , 1998; Trias et al. , 2000; Park et al. , 1999 ).
5. Economic assessment.
Laparoscopic splenectomies generally cost less than open splenectomies. Park et al. (1999) reported an average of USD 3311 as opposed to USD 3861 for his patients. In this series, the cost of a splenectomy without complications was USD 2794 with laparoscopy and USD 3362 with open surgery. In case of conversion, the cost came to USD 3443. These figures were similar in other series ( Glasgow et al. , 1997; Watson et al. , 1997 ). For Hashizume et al. (1996) , the cost of laparoscopic splenectomies is significantly lower (p<0.001). The only series with opposing results is that of Smith et al. (1996) , which reports a higher cost for laparoscopy.
6. Conclusion.
Laparoscopic splenectomy has become the treatment of choice for certain hematological diseases such as ITP and hemolytic anemias. With a related mortality rate close to zero and a low rate of morbidity, it decreases the hospital stay by half and increases the quality of life of the patients who undergo surgery by reducing the occurrence of postoperative ileus and the intake of analgesics. According to most reports, the cost for LS is lower than for open surgery.
Results are more debatable in case of neoplastic splenomegaly, in obese patients and when the surgeon is not experienced. Splenomegaly is no longer considered a contraindication, thanks to the development of the hand-assisted technique.
Laparoscopy is contraindicated in case of portal hypertension.
The lateral positioning of the patient, or “hanging spleen technique”, along with the use of mechanical stapling of the pedicle are the key technical points of LS.

|